

Updated: 2024-01-10

Li Xiangyang, a doctoral student at the School of Chemistry and Chemical Engineering of the Beijing Institute of Technology (BIT), has recently published a research paper titled "All-Direct Laser Patterning Zinc-Based Microbatteries" in the top international journal Advanced Functional Materials as the first author (DOI: 10.1002/adfm.202314060). BIT is the first author affiliation for the paper, and Zhao Yang, a special researcher at the School of Chemistry and Chemical Engineering at BIT, is the corresponding author.
High-performance shape-customizable micro-power sources with structural flexibility and adaptability to various shapes are gaining increasing attention in the next generation of flexible electronic products. They can provide unique advantages for applications in micro-robots, wearable sensors, and implantable medical devices. Among these micro-power sources, zinc-ion microbatteries based on aqueous systems have attracted wide attention due to their high theoretical capacity of 819 mAh g-1, low redox potential of -0.76 V vs SHE, low cost, and high safety. Currently, the construction methods for zinc-ion microbatteries typically involve electrochemical deposition methods and physical methods (mask-assisted filtration method, photolithography, screen printing, and 3D printing). However, regardless of the method used, achieving smaller and more flexible planar micro zinc-ion batteries usually requires multiple stringent and complex procedures, limiting existing micro-energy devices to simplified interdigital forms. Therefore, to meet the diverse needs of intelligent microelectronic systems in different scenarios, it is crucial to develop simple, economical, high-resolution processing techniques to manufacture high-precision and shape-controllable high-performance microbatteries, making them more widely applicable in practical applications.
Against this background, researchers have, for the first time, used all-direct laser patterning technology (DPL) to construct planar quasi-solid-state Zn//MnO2 microbatteries (MBs) in situ on a flexible substrate. This processing technology can not only achieve the manufacturing of mask-free and efficiently arbitrary geometric patterns, such as circles, paper-cuttings, tropical fish, and house shapes, but also spontaneously generate oxygen vacancies in microelectrode materials, further increasing the electrochemical activity sites of the electrode material and improving the battery's electrochemical performance. The prepared Zn//MnO2 microbattery has a high areal capacity of 0.57 mAh cm-2 and an energy density of 0.75 mWh cm-2, surpassing most aqueous Zn-based microbatteries and Li/Na-based MBs. More importantly, these microbatteries can be easily integrated into on-chip microelectronic systems as built-in power sources, and are highly compatible with various sensing functions, accurately monitoring signals such as wrist bending, pulse, temperature, and humidity.
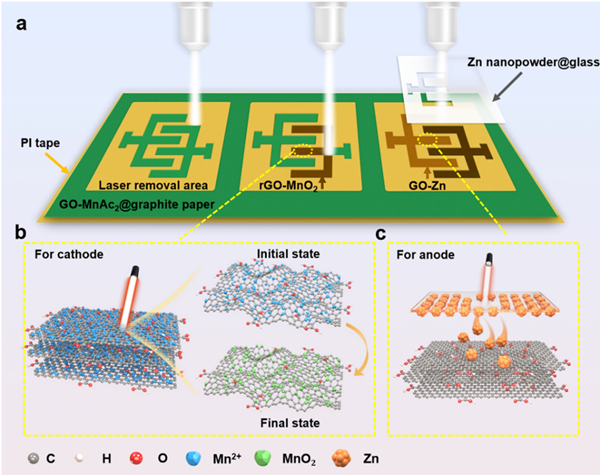
Figure 1. Preparation and schematic diagram of planar GO-Zn//rGO-MnO2 microbatteries (MBs)
The schematic diagram of the full laser processing manufacturing of the planar interdigital electrode of the microbattery is shown in Figure 1. This full laser processing technology has several advantages, including achieving high-resolution electrode patterns by removing material from specified areas and preparing highly active cathode and anode materials.
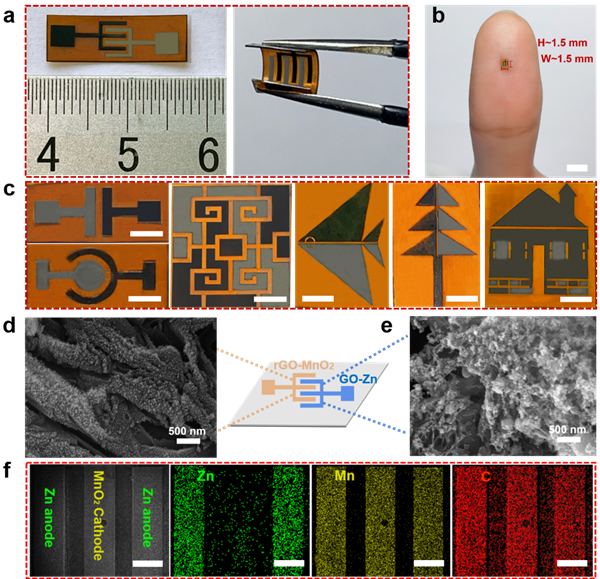
Figure 2. Electron micrograph of microbatteries and morphological characterization of microelectrodes
The overall size of a single microbattery is approximately 0.6 cm2. It is observed that even in a bent state, the microbattery can maintain its structural integrity without detaching from the substrate, demonstrating excellent flexibility. Due to the high precision of the laser, the minimum unit area of the microbattery can reach ~2.3 mm2 (~1.5 mm wide, ~1.5 mm high), with a finger’s width and electrode gaps of just ~160 µm and ~100 µm, respectively. Additionally, it can achieve the direct mask-free micro-pattern preparation of arbitrary geometric shapes. Scanning electron microscope images and mapping images further prove the successful construction of microelectrodes.
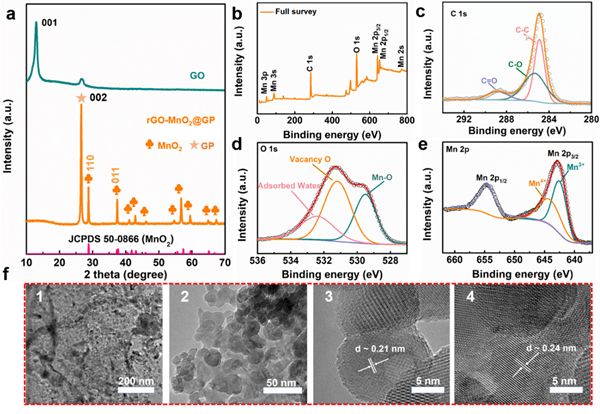
Figure 3. Physical characterization of electrode materials
In the high-resolution spectrum of O 1s, in addition to the Mn-O bond (529.5 eV), two additional peaks at 531.2 eV and 532.4 eV are distinguished, corresponding to oxygen vacancies and adsorbed water molecules, respectively. This indicates the successful introduction of oxygen-related electrochemically active defects in positive electrode materials during laser processing.
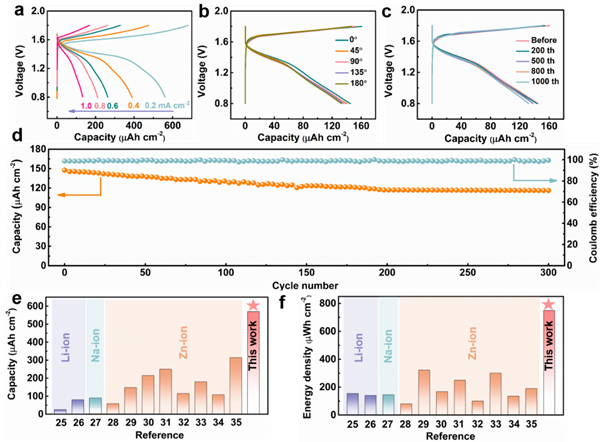
Figure 4. Electrochemical performance of microbatteries
The constructed Zn//MnO2 microbattery exhibits a high capacity of 0.57 mAh cm-2 at a current density of 0.2 mA cm-2, with an energy density of 0.75 mWh cm-2. Its performance surpasses that of most aqueous zinc-ion microbatteries and lithium/sodium microbatteries.
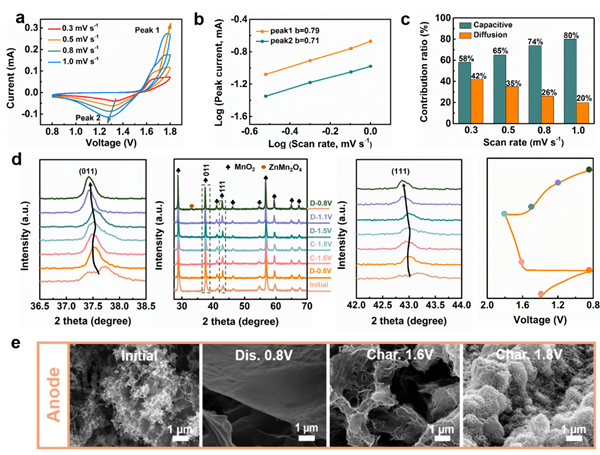
Through reaction kinetics analysis, it is evident that the reaction kinetics in Zn//MnO2 are controlled by capacitive behavior. Further XRD and SEM images of the electrodes during charging and discharging elucidate the energy storage mechanism of the battery: the positive electrode undergoes an insertion/extraction process of Zn2+, while the negative electrode experiences a Zn2+ electroplating/stripping process.
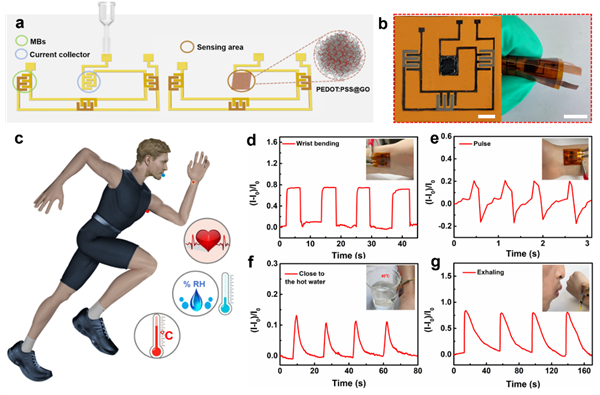
Due to the editability, non-contact nature, and high efficiency of full laser processing technology, a flexible multifunctional integrated system that does not require an external power source can be rapidly written directly onto a chip. This integrated system can continuously monitor human physiological signals (strain, temperature, and humidity signals) to achieve personalized healthcare.
In summary, this work proposes a simple and effective full laser patterning strategy to construct microbatteries with high electrochemical performance. This strategy eliminates the need for complex manufacturing procedures and time-consuming post-processing associated with traditional microbattery manufacturing. By properly introducing oxygen vacancies into the MnO2 positive electrode, the prepared Zn//MnO2 microbattery has a high areal capacity of 0.57 mAh cm-2, surpassing most planar aqueous Zn-ion microbatteries and Li/Na microbatteries. Furthermore, to further demonstrate the versatility of this strategy, a fully flexible integrated system that continuously monitors human physiological signals has been constructed on a single flexible substrate, achieving personalized healthcare. This work provides new opportunities for the manufacturing of scalable microbatteries and the integration of different functional devices on a single substrate, with wide applications in energy storage, microelectronics, and sensing.
Paper link: https://onlinelibrary.wiley.com/doi/10.1002/adfm.202314060
About the author:
Zhao Yang is a special researcher and doctoral supervisor at the BIT School of Chemistry and Chemical Engineering. Zhao has published over 50 SCI papers as corresponding author (or first author), including Nat. Commun., Sci. Adv., Angew. Chem. Int. Ed., J. Am. Chem. Soc., Adv. Mater., Energy Environ. Sci., ACS Nano, and Adv. Funct. Mater., with a cumulative total of more than 100 SCI papers and over 10,000 citations. His four papers have also been selected as ESI Highly Cited Papers (Top 1%). He has led projects such as the National Natural Science Foundation of China General Program and the Beijing Natural Science Foundation. He has also participated in various national major basic research and development (973) projects, as well as key research and development projects. Zhao was also selected as the 2017 Beijing Natural Science Foundation Outstanding Young Talent and listed as a Global "Highly Cited Scientist" by Clarivate Analytics in 2022. He also serves as a junior editor for the international journal Nano Research Energy, an editorial board member for Nanomaterials and Frontiers in Catalysis, as well as a guest editor for the special issue of Nanoscience in the journal Frontiers in Chemistry.














